Anterior cruciate ligament reconstruction

Zoe Russell presents a case study analysing risk factors and considerations for chronological versus outcome-based progression in rehabilitation for a return to running.
A 19 year old male semi-elite football goal keeper presented following an injury to his right knee. The patient, who is right hand and right foot dominant, reported a non- contact injury, valgus rotation mechanism following clearance of a goal attempt towards the end of a training session. He initially reported feeling his knee ‘dislocate’ and reported the knee felt ‘weak and numb’.
Subjective examination
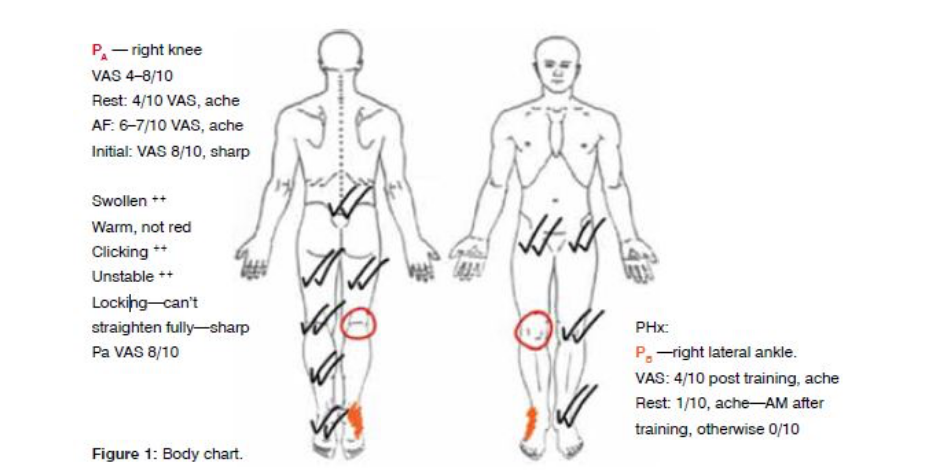
The patient reported constant dull aching pain at rest as identified by region PA in Figure 1, which was worsened by standing, walking (VAS 7/10) and sitting with flexed knee (VAS 6/10). Sharp pain was reported when attempting to extend the knee (VAS 8/10) with a click and locking. Relief was reported with cryotherapy and paracetamol (VAS 4/10). This patient’s subjective reporting of the mechanism and progression of symptoms is consistent with a provisional diagnosis of anterior cruciate ligament (ACL) rupture, with suspicion of meniscal lesion given the locking reported by the patient.
History
The patient works full time as a fruiter and had recently deferred university studies. He denied any history of pelvic, hip and lower limb injury, and reported no history of knee pain. Family history included a paternal ACL injury of the right knee at age 20.
The patient confirmed an interrupted pre-season due to surgical repair of the ulnar collateral ligament of the first metacarpophalangeal joint of his left hand. Subsequently, a second metatarsal stress fracture of the right foot was reported 18 weeks prior to his presenting injury, and a subsequent lateral ligament injury to the right ankle, six weeks prior to sustaining the presenting injury. The patient reported that he continued to play and train at full capacity and did not seek intervention as he did not want to lose his position in the squad. Clanton (2012) advocates the use of clinical testing to assess return to sport (RTS) following ankle injury; given that the patient was receiving no treatment, it is unclear as to whether any range of motion (ROM), neuromuscular and performance deficits influenced development of the knee injury.
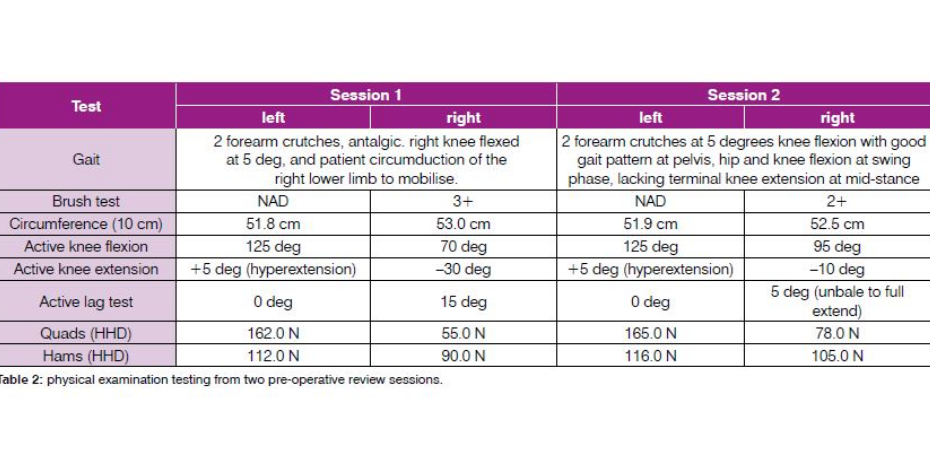
Table 1 outlines relevant physical examination results, validated for the suspicion of ACL rupture (van Eck et al 2013). Assessment was undertaken of the ankle and foot; however, for the purpose of this case study, it will not be discussed further.
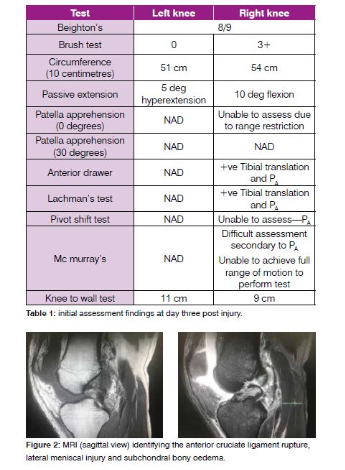
Figure 2 identifies relevant views of the MRI identifying rupture of the ACL, with micro- trabecular injury to the lateral tibial plateau and lateral femoral sulcus, bucket handle tear of the posterior horn of the lateral meniscus, with multidirectional tear and displacement of the medial meniscus. The patient was awaiting the sports physician to present his case to an orthopaedic specialist who works with elite footballers. The patient’s immediate goal was to start rehabilitation for RTS. The patient was committed to having an anterior cruciate ligament reconstruction (ACLR) and meniscal repair as he believed it was his only option to return to semi-elite football.
Management phase 1: pre-surgery Initial management focused on reduction of pain, restoring ROM, reduction of joint effusion, restoration of gait pattern and initiating early quadriceps and hamstrings strength, as this goal of a ‘silent’ knee has demonstrated improved outcomes following ACLR (Cooper & Hughes 2018, van Melick et al 2016, van Grinsven et al 2010). The patient undertook this through cryotherapy, and exercises for ROM, inner range quadriceps, calf and gluteal strengthening, consistent with protocols recommended in the literature (van Grinsven et al 2010).
Education about the nature of the injury, mutual expectations for rehabilitation, progression and outcome measures that would be utilised in rehabilitation, consistent with recommended guidelines from van Melick et al (2016) and van Grinsven et al (2010) reviews, were provided. As a result of the family history of ACL injury, which is a known risk factor (Hewett et al 2015, O’Malley et al 2015), the patient’s family were taught the FIFA 11+ program for injury prevention because it was specific to the patient and his RTS, as well as his siblings risk profile for ACL injury. The FIFA 11+ program is proven effective in reducing ACL rupture incidences (Luiso et al 2018, Silvers-Granelli et al 2015).
Table 2 identifies the changes in the patient’s progress from his first presentation to his pre-surgery assessment of his knee function.
This phase was complicated by the fact that the patient self- administered wall squats to 90 degrees in weight bearing. Consequently, the patient reported increased VAS levels, and this was hypothesised to be a result of bony trabecular injury, which has demonstrated correlation with increased VAS scores with weight-bearing exercise (Papalia et al 2016). It is unclear the role this may have had on the development of quadriceps strength assessed via hand-held dynamometry. The patient self- reported frustration and was referred to a sports psychologist, given the impact of self-efficacy, locus of control and re-injury fear have on RTS following ACLR (Grindem et al 2016). Arden et al (2013) identify clinical screening for maladaptive psychological factors can be undertaken for correlation with RTS; in this case, these measures were administered in the clinic as part of this referral process. Physical impairments in extension ROM and the quadriceps deficits identified in Table 2 are risk factors for post-operative lack of extension ROM, and poor self-reported outcomes at two years (van Melick et al 2016); however, it was hypothesised that the meniscal injuries were responsible for these impairments, rather than failure of pre-surgical management.
Phase 2: surgical reconstruction and meniscal repair
The patient underwent an ACLR using a gracilis-semitendinosus graft, medial and lateral meniscal repair. In the initial six-week period the patient was immobilised non-weight bearing in a splint for protection of the meniscal repair. The orthopaedic specialist was pleased with the surgical approximation of the meniscal injuries and the ACL graft, as reported by the patient. However, the specialist was contacted to obtain an operative report and discuss the perioperative findings, as recommended by van Melick and colleagues (2016) for physiotherapists following ACLR to understand the status of the knee as identified via arthroscopy.
Phase 3: postoperative care
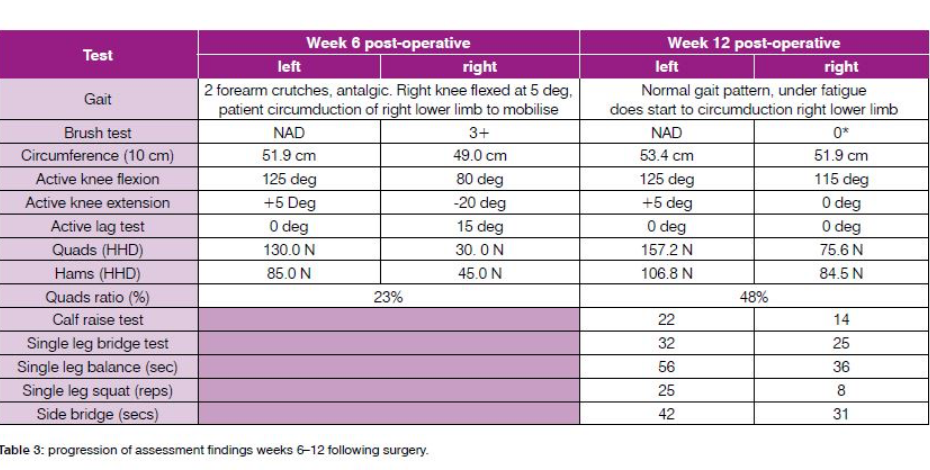
The patient returned to the clinic for review after his six-week review in July 2018. Table 3 outlines the patient’s progression from week six to 12 following ACLR. Performance criteria assessment was based on the recommendations of the Melbourne ACL Rehabilitation Guide (Cooper & Hughes 2018). As demonstrated in
Table 3, the patient achieved the goals of no joint effusion, and while the patient achieved 25 single leg bridge, the patient’s achievement was not greater than 85 per cent of the non-injured side and was deemed to not pass this criterion (Freckleton et al 2013). Additionally, strength measures identified in table 3 demonstrated that the patient had not achieved acceptable performance to progress from the strength and neuromuscular phase to running, jumping and landing tasks (Cooper & Hughes 2018).
Twelve weeks has been identified as chronological criteria to initiate running in a number of protocols; however, less than 20 per cent of these protocols utilise strength or performance criteria prior to initiating a return to running (RTR) (Rambaud et al 2018). Evidence suggests that running should be facilitated once performance criteria have been achieved; based on quadriceps ratio and the outcomes of strength tests outlined in Table 3, the patient was advised for further strengthening and neuromuscular training and to refrain from running until these goals were achieved.
Discussion
This case outlines a typical presentation of knee pain that presents to sports physiotherapists in the clinic. Non-contact with valgus and external rotation force is the most common mechanism of ACL rupture, with the dominant foot most commonly injured (Luiso et al 2018). Provisional diagnosis of ACL rupture with suspicion of meniscal injury was made using subjective and validated physical examination tests, which are still valid despite the sensitivity of 86.2 per cent, and a specificity of 90.7 of MRI in the investigation of ACL and meniscal injuries (Crawford et al 2007, van Eck et al 2013).
Non-modifiable factors such as the patient’s age, generalised joint laxity, and family history of ACL place this patient at greater risk of ACL rupture (Smith et al 2012a, Smith et al 2012b). This patient’s history of stress fracture and lateral ankle sprain are significant, given that lateral ankle sprains on the ipsilateral limb have been identified as a risk factor for ACL injury (Kramer et al 2007). Suspicion regarding the patient’s neuromuscular status must be considered as both a risk factor and impairment in this case in the development of injury (Smith et al 2012a); however, this cannot be proven in this case because assessment and RTS testing of the ankle was not performed prior to ACL rupture and ACLR. Future cases should ensure that neuromuscular assessment is undertaken following lower limb injury and neuromuscular assessment prior to ACLR, given that comparison of functional tasks can guide clinical decision-making (Cooper & Hughes 2018, Hewett et al 2015, Clanton et al 2012).
Gracilis-semitendinosus grafts have reported higher likelihood of a return to football (Luiso et al 2018) and non-surgical management was not considered in this case, despite similar outcomes to ACLR (Monk et al 2016). In this case, the meniscal injuries, the patient’s age and joint laxity, as well as a desire to return to football, all have a high association with secondary injury and there is evidence advocating early surgical intervention to return to pivoting sports (Oiestad et al 2018, Grindem et al 2016, van Melick et al 2016, Webster et al 2014). In this case, surgical timing was based on surgical availability, not these RTS considerations. Wyatt (2017) reviewed patient and orthopaedic criteria for positive outcomes and identified that orthopaedic specialists with sports fellowship training and younger patients with low BMI are favourable for positive outcomes following ACLR in combined injuries. Given the proven relationship of meniscectomies with degenerative osteoarthritis, poor knee function, and retention of the menisci for load transmission and chondro-protection, the interplay of these factors in this case was favourable; however, clinical review following RTS with KOOS measures in the future is recommended for this patient.
In this case, pre-ACLR strengthening was undertaken, with exhibited improvements in effusion, ROM and strength. Despite this, improvements were not comparable to the non-injured knee and the ‘silent’ knee that is recommended was not achieved (van Grinsven et al 2010). It is currently indeterminate whether two weeks of therapy was enough to influence this patient’s postoperative course given that these measures were improving; however, it is hypothesised that in this case the use of performance criteria negates the influence of the shortened pre-ACLR rehabilitation compared to the literature, as it may be a significant risk factor in chronological progression in this case (Cooper & Hughes 2018, Failla et al 2016, Filbay et al 2016).
Successful RTS in those who undertake ACLR is the desired goal, and this case is no exception. Time-based guidelines have been historically reported in the management of ACLR, with 12 weeks identified as a milestone for RTR, and minimal deviation from this timeframe based on patient characteristics, surgical technique and concomitant injury (Kyritsis et al 2016). A recent review identifies that many of the studies that advocate for chronological progression fail to undertake performance criteria to provide clinical justification for RTR (Rambaud et al 2018). This case is a primary example of a patient whose clinical performance criteria do not correlate with chronological progression for RTR, and it is not appropriate for this patient at 12 weeks based on ROM and strength and performance criteria (Cooper & Hughes 2018, Rambaud et al 2018). As this patient progresses through rehabilitation, performance criteria will be measured, and compared to his pre- season screening data to plan his RTS.
Conclusion
Rambaud et al (2018) identified return to running (RTR) as a crucial element of the RTS continuum following ACLR, and progression through a rehabilitation program influences long-term outcomes with RTS. This particular case demonstrates an individual’s
RTR was not appropriate based on chronological rehabilitation milestones following ACLR. Current literature suggests that RTR and functional progression following ACLR should be individualised and based upon the outcome of a number of functional and strength tests over the course of the rehabilitation program. Future investigation and comparison of secondary injury rates, RTR and RTS, and osteoarthritis rates in patients who undertake performance criteria progression compared to those who have undertaken chronological progression, should be undertaken to confirm best clinical practice.
The patient has consented to being involved in this case study.
Zoe Russell is an APA Sports and Exercise Physiotherapist, and APA Musculoskeletal Physiotherapist working in private practice in Brisbane. She is currently undertaking the sports and exercise specialisation program through the Australian College of Physiotherapists.
References
Ardern, C. L., Taylor, N. F., Feller, J. A., Whitehead, T. S., & Webster, K. E. (2013). Psychological responses matter in returning to preinjury level of sport after anterior cruciate ligament reconstruction surgery. American Journal of Sports Medicine. 41:1549–58.
Bisciotti, G. N., Chamari, K., Cena, E., Carimati, G., & Volpi, P. (2016). ACL injury in football: a literature overview of the prevention programs. Muscles, Ligaments and Tendons Journal. 6 (40): 473 – 479.
Clanton, T. O., Matheny, L. M., Jarvis, H. C., & Jeronimus, A. B. (2012). Return to play in Athletes following ankle injuries. Sports Health. 4 (6): 471 – 474.
Crawford, R., Walley, G., Bridgman, S., & Maffuli, N. (2007). Magnetic resonance imaging versus arthroscopy in the diagnosis of knee pathology, concentrating on meniscal lesions and ACL tears: a systematic review. British Medical Bulletin. 84: 5 – 23.
Cooper, R., & Hughes, M. (2018). The Melbourne ACL Rehabilitation Guide 2.0.
Failla, M. J., Arundale, A. J. H., Logerstedt, D. S., & Snyder – Mackler, L. (2015). Controversies in Knee Rehabilitation: Anterior Cruciate Ligament Injury. Clinics in Sports Medicine. 34 (2): 301 – 312.
Filbay, S. R., Roos, E. W., Frobell, R. B., Roemer, F., Ranstam, J., & Lohmander, L. S. (2017). Delaying ACL reconstruction and treating with exercise therapy alone may alter prognostic factors for 5 – year outcome: an exploratory analysis of the KANON trial. British journal of Sports Medicine. 0: 1 – 9.
Grindem, H., Snyder – Mackler, L., Moksnes, H., Engebretsen, L., & Risberg, M.A. (2016). Simple decision rules can reduce re – injury risk by 84% after ACL reconstruction: the Delaware – Oslo ACL cohort study. British Journal of Sports Medicine.50: 804 – 808.
Hewett, T. E., Myer, G. D., Ford, K. R., Paterno, M. V., & Quatman, C. E. (2016). Mechanisms, Prediction, and Prevention of ACL injuries: Cut Risk with Three Sharpened and Validated Tools. Journal of Orthopaedic Research. V: 1842 – 1855.
Kramer, L. C., Denegar, C. R., Buckley, W. E., & Hertel, J. (2007). Factors associated with anterior cruciate ligament injury: history in females. Journal of Sports Medicine and Physical Fitness. 47 (4): 446 – 454.
Kyritsis, P., Bahr, R., Landreau, P., Miladi, R., & Witvrouw, E. (2016). Likelihood of ACL graft rupture: not meeting six clinical discharge criteria before return to sport is associated with a four times greater risk of rupture. British Journal of Sports Medicine, 50: 946 – 951.
Luiso, F., Tromponi, C., Pozza, P., Cavazza, E., Vecchini, E., & Ricci, M. (2018). Anterior cruciate ligament injury in amateur football players: risk factors and return to sport after surgical reconstruction. Sports Sciences for Health. 14: 365 – 370.
Monk, A. P., Davies, L. J., Hopewell, S., Harris, K., Beard, D. J., & Price, A. J. (2016). Surgical Versus conservative interventions for treating anterior cruciate ligament injuries. Cochrane Database of Systematic Reviews. 4.
O’Malley, K., Rubinstein, A., & Postma, W. (2015). Anterior Cruciate Ligament Injury. Current understanding of Risk Factors. Orthopaedics and Rheumatology. 1(3): 1 – 8.
Oiestad, B. E., Holm, I., & Risberg, M. A. (2018). Return to Pivoting sport after ACL reconstruction: association with osteoarthritis and knee function at the 15 – year follow up. British Journal of Sports Medicine. 52: 1199 – 1204.
Poulson, E., Concalves, G. H., Roos, E. M., Thorlund, J., & Juhl, C. B. (2017). Quantifying the risk of developing knee osteoarthritis following knee injury – a systematic review and meta-analysis. Osteoarthritis and Cartilage. 25: S363.
Rambaud, A. J. M., Ardern, C. L., Thoreux, P., Regnaux, J. P., & Edouard, P. (2018). Criteria for return to running after anterior cruciate ligament reconstruction: a scoping review. British Journal of Sports Medicine. 0: 1 – 9.
Silvers – Granelli, H., Mandelbaum, B., Adenii, O., Insler, S., Bizzini, M., Pohlig, R., Junge, A., Snyder – Mackler, L., & Dvorak, J. (2015). Efficacy of the FIFA 11 + Injury prevention program in the collegiate Male Soccer player. American Journal of Sports Medicine. 43 (11): 2628 – 2637.
Smith, H. C., Vacek, P., Johnson, R. J., Slauterbeck, J. R., Hashemi, J., Shultz, S., & Beynnon, B. D. (2012). Rick Factors for Anterior Cruciate Ligament Injury: A review of the Literature – Part 1: Neuromuscular and Anatomic Risk . Sports Health. 4 (1): 69 - 78.
Smith, H. C., Vacek, P., Johnson, R. J., Slauterbeck, J. R., Hashemi, J., Shultz, S., & Beynnon, B. D. (2012). Rick Factors for Anterior Cruciate Ligament Injury: A review of the Literature – Part 2: Hormonal, Genetic, Cognitive Function, Previous Injury and Extrinsic Risk Factors. Sports Health. 4 (2): 155 – 161.
Van Eck, C. F., can den Bekerom, M. P. J., Fu, F. H., & Kerkhoffs, G. M. (2013). Methods to diagnose acute anterior cruciate ligament rupture: a meta – analysis of physical examinations with and without anaesthesia. Knee Surgery, Sports Traumatology, Arthroscopy. 21 (8): 1895 – 1903.
Van Grinsven, S., van Cingel, R. E. H., Holla, C. J. M., & van Loon, C. J. M. (2010). Evidence – based rehabilitation following anterior cruciate ligament reconstruction. Knee Surgery, Sports Traumatology, Arthroscopy. 18: 1128 – 1144.
Van Melick, N., van Cingel, R. E. H., Brooijmans, F., Neeter, C., van Tienen, T., Hullegie, W., & Nijhuis – van der Sanden, M. W. G. (2016). Evidence – based clinical practice update: practice guidelines for anterior cruciate ligament rehabilitation based on a systematic review and multidisciplinary consensus. British Journal of Sports Medicine. 50: 1506 – 1515.
Walden, M., Hagglund, M., Magnusson, H., & Ekstrand, J. (2016). ACL injuries in men’s professional football: a 15 year prospective study on time trends and return to play rates reveals only 65% of players still play at the top level 3 years after ACL rupture. British Journal of Sports Medicine. 50: 744 – 750.
Webster, K.E., Feller, J. A., Leigh, W. B., & Richmond, A. K. (2014). Younger patient are at increased risk for graft rupture and contralateral injury after anterior cruciate ligament reconstruction. The American Journal of Sports Medicine. 42(3): 641 – 647.
Wyatt, R. W. B., Inacio, M. C. S., Liddle, K. D., & Maletis, G. B. (2013). Factors associated with meniscus repair in patients undergoing anterior cruciate ligament reconstruction. The American Journal of Sports Medicine. 41 (12): 2766 - 2771
Zbrojkiewicz, D., Vertullo, C., & Grayson, J. E. (2018). Increasing rates of anterior cruciate ligament reconstruction in young Australians, 2000 – 2015. Medical Journal of Australia. 208 (8): 354 – 358.
© Copyright 2025 by Australian Physiotherapy Association. All rights reserved.